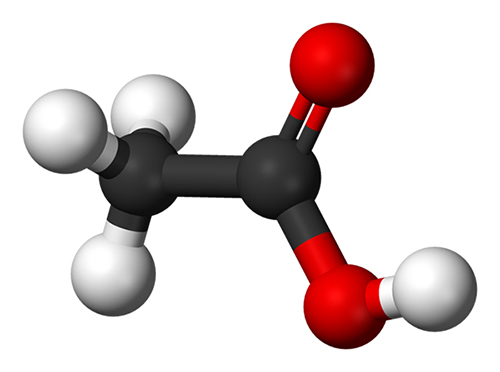
Acetic acid, also called Glacial acetic acid,it is an organic compound with the chemical formula CH3COOH, which is the main component of vinegar.Acetic acid is a colorless and transparent liquid with a pungent odor, soluble in water, ethanol, ether, glycerin, and insoluble in carbon disulfide. It is generally present in many plants in nature in free or ester form.The Glacial acetic acid can be divided into food grade glacial acetic acid and industrial grade glacial acetic acid.
Uses of glacial acetic acid in industry:
Acetic acid is a staple chemical product and one of the most important organic acids. Mainly used in the production of acetic anhydride, acetate and cellulose acetate.
Acetates formed from lower alcohol are excellent solvents and are widely used in the paint industry. Acetic acid is also a commonly used organic solvent because it dissolves most organics.
Acetic acid can be used in some pickling and polishing solutions, as a buffer in weak acid solutions, as an additive in semi-bright nickel plating electrolytes, and in passivation solutions of zinc and cadmium to improve the binding force of passivation films, It is often used to adjust the pH of weakly acidic baths.
Acetic aicd is used in the production of acetate, such as manganese, sodium, lead, aluminum, zinc, cobalt and other metal salts, widely used as catalyst, fabric dyeing and leather tanning industry auxiliaries; lead acetate is paint color lead white; Lead tetraacetate is an organic synthetic reagent.
Acetic acid can also be used as analytical reagents, organic synthesis, synthesis of pigments and pharmaceuticals.
The use of glacial acetic acid in the food industry:
In the food industry, acetic acid is used as an acidulant, flavor enhancer and flavoring agent to make synthetic vinegar, adding various flavoring agents, the flavor is similar to that of alcohol, the manufacturing time is short, and the price is cheap. As a sour agent, it can be used in compound seasonings, in the preparation of vinegar, canned food, jelly and cheese, and used in moderation according to production needs. It can also be used as a flavor enhancer for composing wine.
Glacial acetic acid has dangerous properties: it can react violently with oxidants, and react violently with sodium hydroxide and potassium hydroxide. Corrosive to metals when diluted.
Acetic acid in higher concentrations is corrosive and can cause burns to the skin, permanent blindness of the eyes, and inflammation of the mucous membranes, requiring proper protection.
Glacial Acetic Acid Electrostatic Action: Possibly a polymerization hazard.
Post time: Jul-12-2022